
Medical devices
The medical device industry is a crucial segment of the healthcare sector, involved in the design, manufacture, and marketing of devices that diagnose, prevent, monitor, treat, or alleviate diseases and health conditions.
The medical device industry is a crucial segment of the healthcare sector, involved in the design, manufacture, and marketing of devices that diagnose, prevent, monitor, treat, or alleviate diseases and health conditions. This industry includes a wide range of products, from simple bandages and spectacles to complex programmable pacemakers and sophisticated imaging systems.
Key ISO Standards Relevant to the Medical Device Industry
ISO 13485: Medical Devices – Quality Management Systems
ISO 13485 is the principal standard for quality management systems in the medical device industry. It is designed to work throughout the life cycle of a medical device, from initial conception to production and post-production, including final decommission and disposal. This standard focuses on what manufacturers must do to ensure their devices consistently meet customer and regulatory requirements.
ISO 14971: Medical Devices – Application of Risk Management to Medical Devices
ISO 14971 provides a thorough framework for manufacturers to identify hazards associated with medical devices, including in vitro diagnostic (IVD) medical devices, estimate and evaluate associated risks, control these risks, and monitor the effectiveness of the controls. The purpose of risk management is to ensure that the medical device is as safe as possible, and it plays a crucial role in the product life cycle.
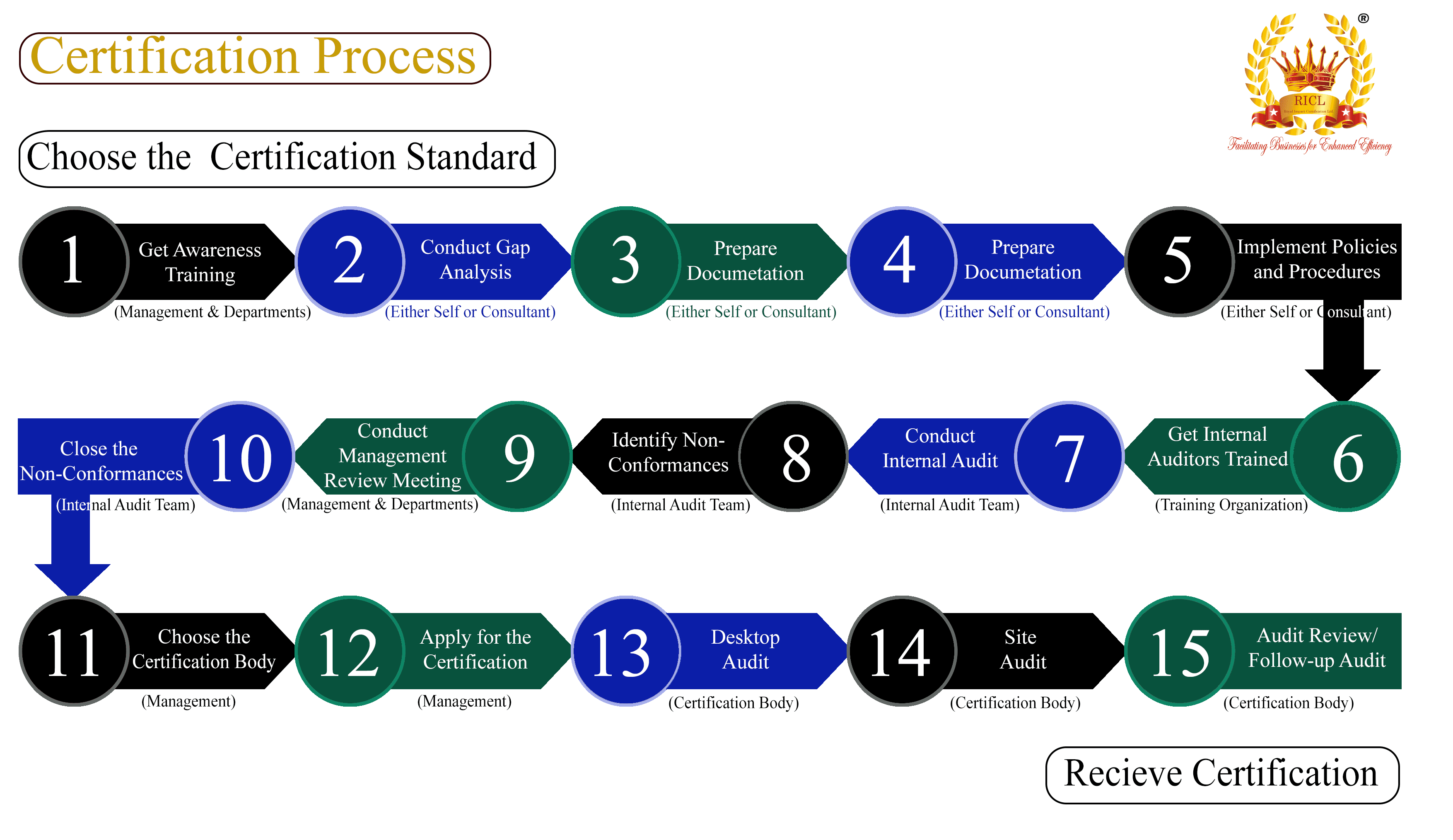
Benefits of ISO Certification in the Medical Device Industry
Enhanced Product Quality and Safety: Adherence to ISO standards helps ensure that medical devices are manufactured under controlled conditions, resulting in higher quality and safer products.
Regulatory Compliance: Many countries require medical device manufacturers to meet ISO 13485 as a part of their regulatory compliance efforts. Compliance with ISO 14971 is also seen as best practice for meeting international safety requirements.
Improved Credibility with Stakeholders: Certification can enhance a company’s reputation and can be a significant competitive advantage, signaling to customers, suppliers, and regulators that the company takes quality seriously.
Operational Efficiency: Implementing ISO standards helps streamline production processes, reduce waste, and lower costs by improving efficiency.
[formsapp id=”662f63f6c4dd546c9916c3db”]
Related Standards
The IT industry is governed by several ISO standards that help ensure quality, safety, security, environmental responsibility, and overall efficiency. Here are some key ISO standards related to the transport and logistics industry:

ISO 13485
Quality Management For Medical Device
LATEST BLOGS
ISO 10002 Enhancing Customer Satisfaction and Complaint Handing Introduction ISO 10002 is an international standard that provides guidelines for the process of …
Who benefits from ISO 50001 Certification for energy performance? ISO 50001 Certification for energy performance benefits a wide range of stakeholders, including …
What ISO 17025 Certification ensures in Laboratory testing? ISO 17025 Certification ensures a high standard of competence and reliability in laboratory testing …



